
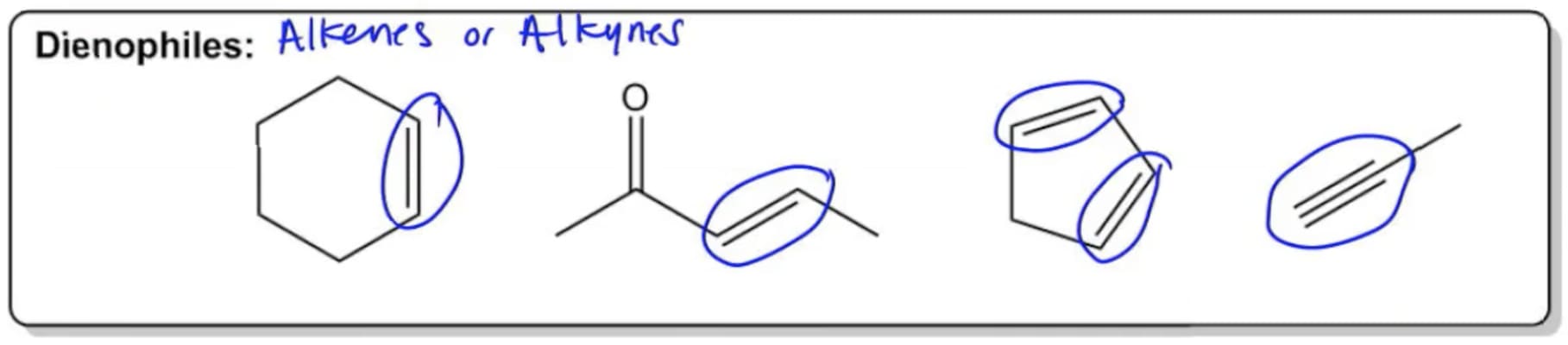
Making a short summary, we can see that depending on the structure of the diene, the 1,2 or 1,4-product is obtained. This time, we can first try the electron-flow method: Let’s also look at an example where the electron-donating group of the diene is on carbon number 1 (1-substituted diene): What if the CH 3O- is on the other carbon? Once you have determined the correct alignment, you can now draw the actual mechanism. Notice again that this is not the mechanism of the Diels-Alder reaction! It is only to quickly predict the major regioisomer of the product. The correct alignment is the one that supports the electron flow from the electron-donating diene substituent to the electron-withdrawing group of the dienophile ( electron-flow method): Simply place the molecules next to each other and draw the curved arrows connecting the first two carbons of the diene and the dienophile You can also predict the major regioisomer of a Diels-Alde reaction without drawing the resonance forms. We are only doing this to predict the major product by connecting the most electron-rich carbon of the diene to the most electron-deficient carbon of the dienophile. Remember that the Diels-Alder reaction follows a concerted mechanism and there is no (ionic) intermediates involved.
The first one is to draw the resonance structures of the diene and dienophile placing the formal charges on the terminal atoms and align the molecules next to each other pairing the opposite charges: You have two ways to determine the proper alignment of the diene and the dienophile. The most electron-rich carbon of the diene reacts with the mot electron-deficient carbon of the dienophile. This regioselectivity is a result of the electron distribution in the diene and the dienophile. How do I figure this out for any Diels-Alder reaction? It turns out that the 1,4-product is the major regioisomer which means that the diene and dienophile follow alignment B in the transition state of the reaction: And the question is which of these regioisomers is the major product? This is the regiochemistry or the regioselectivity of the Diels-Alder reaction. You can simply flip one of the reactants upside down to better visualize the formation of two isomers: These isomers are formed as a result of the two possible orientations (A and B) that the diene and dienophile can have.

What is interesting here is that the first reaction produces only one product (we will disregard the stereochemistry for now), while the second one can give a mixture of two regional (constitutional) isomers. The first one is a reaction between a symmetrical diene and a dienophile and the second one is a reaction of an unsymmetrical diene and a dienophile. The precoated MWCNTs can also promote the CVD reaction of pyrolysis gas in char layers during ablation, decreasing the porosity of char layers, which can increase the particle erosion resistance of insulation.Let’s compare the following two Diels-Alder reactions: The char layers of MWCNTs reinforced formulas are more complete than basic formula, indicating that MWCNTs can improve char layer structure under particle erosion condition. Charring ablation rate of the formula with 10phr 1 precoated MWCNTs is 44.3% lower than that of basic formula and 41.6% lower than that of the formula with 3phr MWCNTs which has the same pure MWCNTs content. Experimental results show that precoated MWCNTs can not only improve thermal insulation performance but also significantly increase particle erosion resistance of ethylene propylene diene monomer insulation materials. Moreover, their ablation performances under simulated overload ablation conditions were tested and compared. Further, ethylene propylene diene monomer insulations reinforced with different contents of MWCNTs and precoated MWCNTs were prepared. In this study, MWCNTs were precoated with pyrolytic carbon via chemical vapor deposition to reduce their thermal conductivity. Multi-walled carbon nanotubes (MWCNTs) can improve erosion resistance of insulations but at the cost of reduced thermal insulation performance. Condensed particles in the internal flow field of a solid rocket motor can cause serious erosion to thermal insulations under overload flight condition.
